Introduction
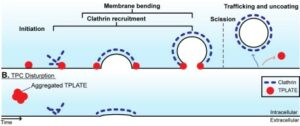
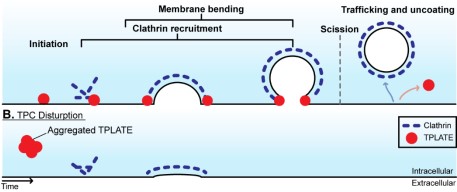
While plant CME is actin independent, the authors identify that the 4 evolutionarily ancient octameric TPLATE complex mediates membrane bending against high 5 turgor pressure in plant clathrin-mediated endocytosis.
Clathrin-mediated endocytosis (CME) is a critical eukaryotic cellular process that regulates a 33 wide range of physiological processes, for example, mediating the internalization of receptors 34 and transporters. During CME, a small area of the plasma membrane (PM) invaginates into 35 the cell forming a spherical vesicle against intracellular forces that oppose membrane 36 deformation, like turgor pressure. The mechanisms driving this process in mammalian and 37 yeast systems have been the subject of extensive study for the better part of 5 decades, which 38 has led to the identification of key proteins that provide the force required to overcome these 39 forces.
Summary
Clathrin-mediated endocytosis in plants is an essential process but the underlying mechanisms are poorly understood, not least because of the extreme intracellular turgor pressure acting against the formation of endocytic vesicles. In contrast to other models, plant endocytosis is independent of actin, indicating a mechanistically distinct solution. Here, by using biochemical and advanced microscopy approaches, we show that the plant-specific TPLATE complex acts outside of endocytic vesicles as a mediator of membrane bending. Cells with disrupted TPLATE fail to generate spherical vesicles, and in vitro biophysical assays identified protein domains with membrane bending capability. These results redefine the role of the TPLATE complex as a key component of the evolutionarily distinct mechanism mediating membrane bending against high turgor pressure to drive endocytosis in plant cells. One Sentence Summary While plant CME is actin independent, we identify that the evolutionarily ancient octameric TPLATE complex mediates membrane bending against high turgor pressure in plant clathrin-mediated endocytosis.
Publication source
- Source of publication: BioRxiv
- Paper title: The TPLATE complex mediates membrane bending during plant clathrin-mediated endocytosis. Johnson, A., Dahhan, D. A., Gnyliukh, N., Kaufmann, W. A., Zheden, V., Costanzo, T., Mahou, P., Hrtyan, M., Wang, J., Aguilera-Servin, J., van Damme, D., Beaurepaire, E., Loose, M., Bednarek, S. Y., & Friml, J. (2021). Published.