Introduction
Recently, microfluidics and 3D cell culture technologies have merged, to increase the potential of in vitro modeling, in order to get even closer to human physiology. Control of the cell microenvironment by microfluidics in a 3D cell culture enables the mechanical control of the cellular shape, physical and chemical characteristics, shear stress, substratum rigidity, temperature, and gas gradient to be studied in a physio-mimetic manner.
Microfluidic devices are now used with 3D cell culture in two primary areas: fundamental research and drug development. In the aim of providing better physiology mimicking models for drug development, 3D liver models development is critical for toxicity, metabolism & excretion studies. The liver has a complex architecture (micro zonation) and different differentiation stages that need to be recapitulating.
The development of these technologies will have a huge impact on healthcare as a solution for many currently untreatable diseases: heart failure, kidney failure, genetic disorders, orphan diseases, cancer…
Abstract
The authors state that “The development of livers-on-a-chip aims to provide pharmaceutical companies with reliable systems to perform drug screening and toxicological studies. To that end, microfluidic systems are engineered to mimic the functions and architecture of this organ.
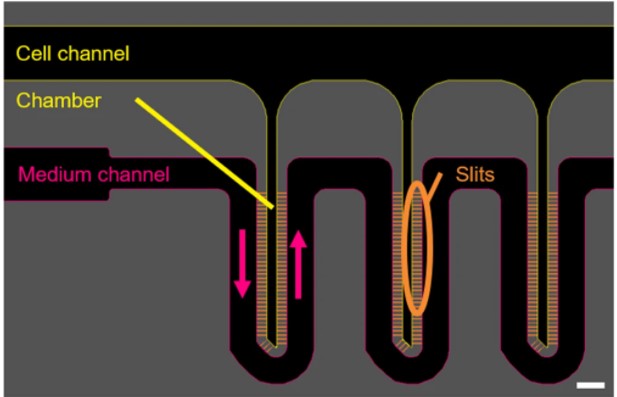
In this context we have designed a device that reproduces series of liver microarchitectures, each permitting the 3D culture of hepatocytes by confining them to a chamber that is separated from the medium conveying channel by very thin slits. We modified the structure to ensure its compatibility with the culture of hepatocytes from different sources. Our device was adapted to the migratory and adhesion properties of the human HepaRG cell line at various stages of differentiation.
Using this device, it was possible to keep the cells alive for more than 14 days, during which they achieved a 3D organization and acquired or maintained their differentiation into hepatocytes. Albumin secretion, as well as functional bile canaliculi, were confirmed on the liver-on-a-chip. Finally, an acetaminophen toxicological assay was performed. With its multiple micro-chambers for hepatocyte culture, this microfluidic device architecture offers a promising opportunity to provide new tools for drug screening applications.”
Source:
Boul M, Benzoubir N, Messina A, Ghasemi R, Mosbah IB, Duclos-Vallée JC, Dubart-Kupperschmitt A, Le Pioufle B. A versatile microfluidic tool for the 3D culture of HepaRG cells seeded at various stages of differentiation. Sci Rep. 2021 Jul 7;11(1):14075. DOI: 10.1038/s41598-021-92011-7. PMID: 34234159; PMCID: PMC8263583.


