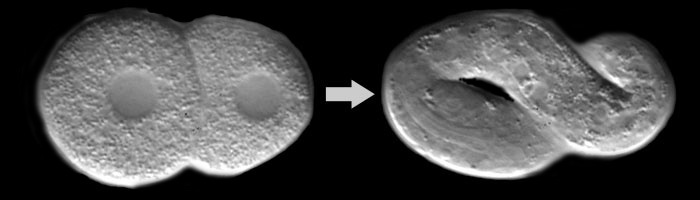
The importance of chiral rotatory movements of the actomyosin cortex, involve in cellular rearrangements, during C. elegans embryo development.
Stephan W. Grill et al. published an article revealing the importance of chiral rotatory movements of the actomyosin cortex in cell position during development of Caenorhabditis elegans embryo. They highlight mechanisms by which rotatory actomyosin flows can specify and reorient the cell division axis. Chiral counter rotating flows drive the AB lineage spindle skew and the reorientation events, giving to them an importance in morphogenesis. This important discovery may give rise ideas about tissue homeostasis and regeneration.
Ultra fast temperature shift device for in vitro experiments under microscopy
Abstract
“Proper positioning of cells is important for many aspects of embryonic development, tissue homeostasis, and regeneration. A simple mechanism by which cell positions can be specified is via orienting the cell division axis. This axis is specified at the onset of cytokinesis, but can be reoriented as cytokinesis proceeds. Rotatory actomyosin flows have been implied in specifying and reorienting the cell division axis in certain cases, but how general such reorientation events are, and how they are controlled, remains unclear. In this study, we set out to address these questions by investigating early Caenorhabditis elegans development. In particular, we determined which of the early embryonic cell divisions exhibit chiral counter-rotating actomyosin flows, and which do not. We follow the first nine divisions of the early embryo, and discover that chiral counter-rotating flows arise systematically in the early AB lineage, but not in early P/EMS lineage cell divisions. Combining our experiments with thin film active chiral fluid theory we identify specific properties of the actomyosin cortex in the symmetric AB lineage divisions that favor chiral counter-rotating actomyosin flows of the two halves of the dividing cell. Finally, we show that these counter-rotations are the driving force of both the AB lineage spindle skew and cell reorientation events. In conclusion, we here have shed light on the physical basis of lineage-specific actomyosin-based processes that drive chiral morphogenesis during development.”
References
FAQ
Proper positioning of cells is noted as being important for many aspects of embryonic development. It is also important for tissue homeostasis and regeneration. A simple mechanism for specifying cell positions is by orienting the cell division axis. This axis is specified when cytokinesis begins. It can, however, be reoriented as the division process proceeds. Rotatory movements of the actomyosin cortex have been implicated in both specifying and reorienting this axis. This process is important for morphogenesis, or the shaping of the organism.
This study set out to address questions about cell division axis reorientation. It aimed to find out how general these reorientation events are. It also sought to understand how they are controlled. The researchers investigated the early development of Caenorhabditis elegans. They determined which of the early embryonic cell divisions display chiral counter-rotating actomyosin flows and which do not. To do this, they followed the first nine divisions of the early embryo. The goal was to understand the physical basis of these lineage-specific processes that drive chiral morphogenesis.
The study discovered that chiral counter-rotating flows arise in a systematic way. These flows were found specifically in the early AB lineage. They were not found, however, in the early P/EMS lineage cell divisions. The researchers combined their experiments with thin film active chiral fluid theory. This approach allowed them to identify specific properties of the actomyosin cortex. These properties, found in the symmetric AB lineage divisions, are what favor the development of these chiral counter-rotating flows. The flows occur in the two halves of the cell as it divides.
The chiral counter-rotating flows were shown to be the driving force for two different events. They are responsible for the AB lineage spindle skew. They also drive cell reorientation events. These rotatory movements of the actomyosin cortex are important in cell positioning during the development of the C. elegans embryo. The mechanisms by which these flows specify and reorient the cell division axis are highlighted in the research. These findings are important because they show how the flows are involved in morphogenesis.