Immobilization of C. elegans during live cell imaging: rational
Although they have proved valuables, all of these procedures show a number of limitations. The use of chemical compounds, induces physiological modifications, which may interfere with the biological question studied. Also, with these techniques, C. elegans worms do not have identical orientation, they may not be easily accessible for further manipulations. While the use of polysterene nanoparticles is easy to set-up, one caveat is that immobilization is dependent on worm growth stages (Kim et al., 2013).

Ultra fast temperature shift device for in vitro experiments under microscopy
Microfluidic device active methods
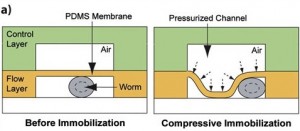
1. The Compressive method. This technique takes advantage of the physical properties of PDMS membrane. In this setting, the microfluidic device contains integrated valves. The C. elegans worm is placed in the bottom flow layer channel, with applied-pressure the membrane is deflected and as a result the worm is physically immobilized. A pressure adjustment allows the use of different worm developmental stages. Pressure applied immobilization may not be compatible with a long immobilization period. Furthermore, the caveats with this technique is that it is not user friendly, it is tricky to generate and the set-up unless one establishes a collaboration with a well trained microfluidic research group. For biologists users, we recommend the use of the passive method, which is more easy to handle.

2. Microfluidic temperature induced immobilization. This method described in Chung et al., 2008 allows a strict immobilization of C. elegans, stopping all internal movement and thus allowing sub-cellular structures imaging. In this setting (see figure) the worm is placed in the bottom flow channel (red), two micro valves control the flow and the worm placement (green), and a temperature-controlled liquid flow channel is placed on top of the worm channel allowing a rapid cool-down of C.elegans temperature. While this method is very useful, it implies a total immobilization of the worm and hence it is not adequate for live-animal video imaging. However, with appropriate devices temperature cool down can be reverse. Also, the use of a worm sorter may prove necessary to get the worms in the same orientation.
C. elegans Immobilization using microfluidic devices: a time-saving strategy!
Here, we present the two main microfluidics strategies to immobilize worms for microscopy imaging : a passive and easy to handle method, in which the worm is trapped in a worm clamp, and an active method in which the worm is immobilized using valves-applied pressure or cold.
The main reason why experimentator will want to use microfluidics is for time-saving considerations and reproducible immobilization conditions. Microfluidics allows a fast handling and consistent immobilization of C. elegans. (San-Miguel A and Lu H, 2013). Moreover, microfluidic immobilization presents several advantages among which unidirectional orientation, no superposition of worms, further manipulation can be performed: such as drug injection or ultra fast temperature control of C. elegans worm environment.
Microfluidic passive method : the worm trap
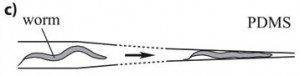
This method is based on a mechanical trapping of C. elegans. The microfluidics chip is composed of parallel microchannels, this is very convenient since it allows high-throughput C. elegans processing. Through action of liquid flow, the worm enters a microchannel, whose width is reduced to a point to that the worm is immobilized (see figure). One advantage of this method is that upon flow reversion, the worm can be retrieved, and drugs can be delivered through the liquid flow and worm can be oriented in the same direction. However, one has to notice that residual movement may occur with this method but it does not appear to be limiting since it can be compensated with appropriate Imaging software (San Miguel and Lu, 2013, wormbook). This method is of particular interest since it can be combined with worm environment manipulation: drugs injection or ultra fast temperature shift.
Conclusion : C.elegans immobilization
Using microfluidic devices for C. elegans immobilization is time saving: with rapid handling, immobilization and imaging time spent per worm experimentation. It allows you to eliminate the use of anesthetics, which perturbed animal physiology. Depending on your needs, you may want to choose an easy to set up method like microfluidic worm traps, or if you need to have a complete block of both external and internal worm movement you’ll find that cooling device may be more suitable.
References
Aufderheide, KJ. (2008). An overview of techniques for immobilizing and viewing living cells. Micron 39, 71–76
Kim E, Sun L, Gabel CV, Fang-Yeng C. (2013). Long-term imaging of Caenorhabditis elegans using nanoparticle-mediated immobilization. PLoS ONE 8(1): e53419
San-Miguel A and Lu H , Microfluidics as a tool for C. elegans research*(September 24, 2013), WormBook, ed. The C. elegans Research Community, WormBook, doi/10.1895/wormbook.1.7.1.
Hulme, S.E., Shevkoplyas, S.S., Apfeld, J., Fontana, W., and Whitesides, G.M. (2007). A microfabricated array of clamps for immobilizing and imaging C. elegans. Lab Chip7, 1515-1523.
Chokshi, T. V., Ben-Yakar, A., and Chronis, N. (2009). CO2 and compressive immobilization of C. elegans on-chip. Lab Chip 9, 151-157.
Chung, K., Crane, M.M., and Lu, H. (2008). Automated on-chip rapid microscopy, phenotyping and sorting of C. elegans. Nat. Methods 5, 637-643.
S. Ben-Aroya, X. Pan, JD. Boeke, and P. Hieter, Making temperature-sensitive mutants, Methods Enzymol. 2010
FAQ
Microfluidic immobilization is presented as a method that provides consistent results and faster handling of C. elegans. This approach is used because other procedures have limitations. For example, the use of chemical compounds can induce physiological modifications. These changes might interfere with the biological question being investigated. Other techniques may result in worms that do not have an identical orientation. They also may not be accessible for subsequent manipulations. Microfluidics is shown to provide unidirectional orientation and prevent the superposition of worms. It also enables further actions, such as drug injection or the application of temperature control.
This method is based on the mechanical trapping of C. elegans. The microfluidic chip contains parallel microchannels, which is convenient for processing many worms. The worm enters a microchannel through the action of liquid flow. The width of this channel is reduced to a point where the worm becomes immobilized. This technique has specific features. Upon reversion of the flow, the worm can be retrieved. Drugs can also be delivered through the liquid flow. Worms can be oriented in the same direction. It is noted that residual movement may occur with this method. However, this movement does not appear to be limiting, as it can be compensated for with appropriate imaging software.
This technique utilizes the physical properties of a PDMS membrane. In this configuration, the microfluidic device contains integrated valves. The C. elegans worm is positioned in the bottom flow layer channel. When pressure is applied, the membrane deflects. As a result of this deflection, the worm is physically immobilized. An adjustment to the pressure permits the use of different worm developmental stages. This method of immobilization may not be compatible with a long period of observation. The source also notes that this technique is not straightforward to implement. It is described as difficult to generate. It is suggested that biologists should use the passive method, as this active one is less simple to manage.
This method, described in a 2008 paper by Chung et al., provides a strict immobilization of C. elegans. It is capable of stopping all internal movement. This makes it suitable for imaging sub-cellular structures. In this device, the worm is placed in the bottom flow channel. The flow and the worm’s placement are controlled by two micro valves. A separate temperature-controlled liquid flow channel is positioned on top of the worm channel. This setup permits a rapid cool-down of the C. elegans temperature. While this method is noted as effective, it results in a total immobilization of the worm. Therefore, it is not considered adequate for live-animal video imaging. The cooling can be reversed with appropriate devices.