Introduction
The use of D. melanogaster as a model organism in developmental biology has been an important tool to understand fundamental biological processes occurring in a complex animal. Following there is a brief description of the main stages during the drosophila life cycle: embryogenesis, larval stage, pupal stage and adult life. During these different stages in the fly life cycle, the structures and organs giving rise to the fly (adult) anatomy are formed.
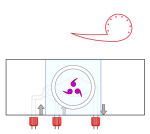

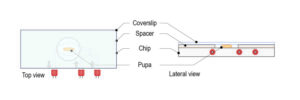
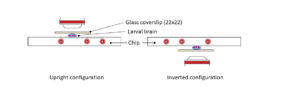
Ultra fast temperature shift device for in vitro experiment under microscopy
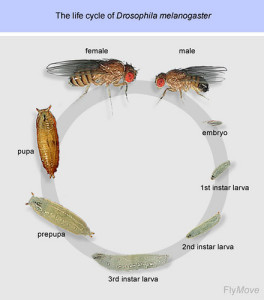
The drosophila life cycle
The life cycle of drosophila, from egg fertilization to adult life, takes about 10 days at 25°C. Drosophila is a model organism particularly used in developmental biology because it is a holometabolous insect, with major morphological differences occurring between larvae and adult animal (metamorphosis) [1]. The drosophila life cycle is comprised of the following developmental stages:
- Embryogenesis: It is a fast process completed 24h after fertilization of the oocyte by the male sperm [2]. From a one cell embryo, a syncytial embryo is rapidly developed. In the early embryo syncytium, rapid DNA replication and nuclear divisions occur, generating up to 5000 nuclei per embryo. Cellularization occurs after migration of nuclei to the periphery of the syncytium, generating the syncytial blastoderm in a process called cleavage. During gastrulation, cells change shape and migrate establishing the mesoderm, endoderm and ectoderm, which are the layers of the future body plan. For more details on cleavage and gastrulation stages see [1] or go to Drosophila embryo live cell imaging.
- Larval stage: There are three larval stages (3 instars) which take altogether about 4 days. During larval growth, most cell types are already differentiated and functional. Therefore, many biological questions can be addressed already at the larval stage. For instance, larvae have been important for neuronal studies, including memory formation. The larval central nervous system consists on only 10000 neurons compared to more than 250000 in the adult fly, providing a simpler model [3]. A molting transition occurs between the different larval stages [1].
- Pupal stage: After encapsulation of the 3d instar larva, pupal stage starts and lasts around 4 days. Many larval structures are lysed and new structures are formed. New structures are generated from the formation of the imaginal disks, developed from larval undifferentiated cells. Imaginal disks will give rise to the adult head, legs, wings, thorax and reproductive apparatus. Some larval structures like the nervous system or gonads are preserved during the pupal stage [1].
- Adult life. Adult fly emerges upon eclosion of the pupal case. Lifespan is around 30 days, although this is variable according to temperature. The high number of eggs layed per female (100 eggs per day), generating a big progeny after genetic crosses, makes drosophila a good genetic model with very easy growth in laboratory conditions
The study of adult flies has led to important discoveries in fundamental biological processes like aging, circadian clocks or behavioural studies.
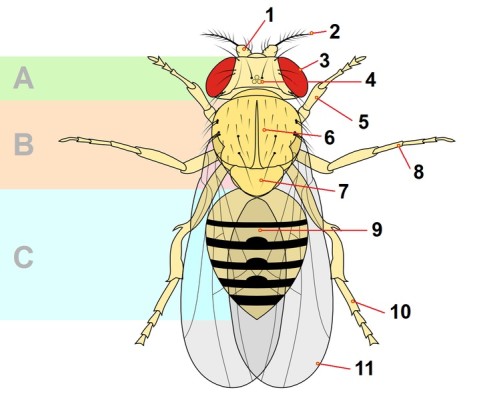
Fly structure (anatomy)
This is a summary of Drosophila melanogaster anatomy. Many studies using drosophila as a model system have focused on the understanding on how fly structures are formed (developmental biology), how they work (neurobiology), and how organs grow as the animal grows.
A collection of images and drawings of the fly anatomy can be found at Flybase [5]. The following link to an anatomical atlas allows to easily identify adult Drosophila structures [6]: http://www.ento.csiro.au/biology/drosophila/melanogaster.html
Some examples of well-studied structures or organs are:
- Compound Eye (3 on the image): it is composed by thousands of cells, with differentiated functions (from photoreception to non-neuronal cells). Multiple studies on patterning and morphogenesis and cell-cell communication have allowed to understand eye function and development [7]
- Antennae (1 and 2 on the image): this is a pair of sensory elements on the head part of the fly. In the second antennal segment (2 in the image) stands a mass of receptor cells, called the Johnston organ. The Johnston organ is the hearing organ of the fruit fly, and it allows flies to sense different mechanosensory stimuli like sound (for instance during courtship) but also wind and gravity. Neuronal signalling from the antenna to the brain allows for behavioural responses to these stimuli [8].
- Proboscis (mouthpart): the mouth part of the fly has an important role in detecting non- volatile compounds, with functions in feeding or detection of toxic compounds, mating (non-volatile pheromones), and egg laying. These receptors though, are not restricted to the mouth and are distributed all along the fly body through hairlike structures. Many studies have focused on gustatory receptors and chemosensation [9].
- Bristles (6 on the image). They are one more component of the Drosophila peripheral nervous system. They are sensory organs placed across the surface of the adult fly. They are mechanoreceptors composed of 4 specialized cells, one of them a neuron. The development of bristles occurs at the late larva and early pupa stages [10]. Bristles are generated from a single cell SOP (sensory organ precursor), what makes them a model structure to understand asymmetric cell division and cell differentiation.
- Ovaries: In the adult female, the largest organ is the ovary. Each ovary contains around 18 ovarioles, where eggs are generated. Oogenesis in the female fly is the process of development of a mature egg (oocyte) from a stem cell. Oogenesis takes place in the ovaries, during about one week. It starts with the budding of the egg chambers (follicles) from the germinarium in the ovary and finishes with the formation of a mature oocyte. Egg chambers are comprised of multiple cells (somatic and germ line) and give rise to a single mature egg. Assymetric cell division and cell fate determinants, together with cell migration, are required for oogenesis [4].
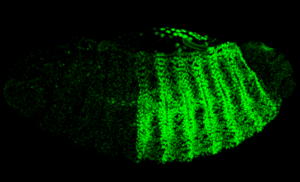
The CherryTemp is designed specifically for temperature control during live imaging of Drosophila embryos and larvae. It provides precise temperature shifts ranging from 5°C to 45°C, crucial for studying temperature-sensitive processes such as development and genetic expression. The system ensures rapid adjustments and stability, making it ideal for manipulating developmental conditions in real-time. It integrates seamlessly with various microscope setups, allowing researchers to maintain optimal conditions for Drosophila studies.

References
- [1] Textbook Principles of Development (5th edition 2015) Lewis Wolpert, Cheryll Tickle, and Alfonso Martinez Arias
- [2] Hales, KG et al. Genetics on the Fly: A Priper on the Drosophila Model System. Genetics.(2015) https://www.ncbi.nlm.nih.gov/pubmed/26564900
- [3] Widmann et al. Odor-taste Learning in Drosophila Larvae. Journal of Insect Physiology (2017) https://www.ncbi.nlm.nih.gov/pubmed/28823531
- [4] Bastock R. and St. Johnston D., Drosophila oogenesis. Current Biology (2009) https://www.ncbi.nlm.nih.gov/pubmed/19081037
- [5] Flybase; Anatomy.
- [6] Anatomical atlas. http://www.ento.csiro.au/biology/drosophila/melanogaster.html#
- [7] Cagan R. Principles of Drosophila Eye Differentiation. Curr Top Dev Biol (2010) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2890271/
- [8] Matsue E. Kamikouchi A. Neuronal encoding of sound, gravity, and wind in the fruit fly. J Comp Physiol A Neuroethol Sens Neural Behav Physiol.(2013) https://www.ncbi.nlm.nih.gov/pubmed/23494584
- [9] Montell C. A taste of the Drosophila gustatory receptors. Curr Opin Neurobiol (2009) https://www.ncbi.nlm.nih.gov/pubmed/19660932
- [10] Furman DP, Bukharina TA. Drosophila mechanoreceptors as a model for studying asymmetric cell division. Int.J.Dev. Biol (2011) https://www.ncbi.nlm.nih.gov/pubmed/21671219
FAQ
The complete life cycle of Drosophila, from egg fertilisation to the adult, takes approximately 10 days when maintained at 25°C. This organism is used in developmental biology research because it is a holometabolous insect. This means major morphological differences happen between the larva and the adult animal, a process known as metamorphosis. The life cycle is composed of several developmental stages. These stages are embryogenesis, the larval stage, the pupal stage, and adult life. During these distinct phases, the structures and organs that make up the adult fly’s anatomy are formed.
Embryogenesis is a fast process that is completed 24 hours after the oocyte is fertilised. A syncytial embryo develops rapidly from a single cell. Inside this early embryo syncytium, quick DNA replication and nuclear divisions take place. This activity generates up to 5000 nuclei per embryo. Nuclei then migrate to the periphery of the syncytium, and cellularization happens. This forms the syncytial blastoderm in a process called cleavage. Following this, during gastrulation, cells will change their shape and migrate. This action establishes the mesoderm, endoderm, and ectoderm, which are the layers for the future body plan.
The larval stage consists of three instars, which last for about 4 days altogether. During larval growth, most cell types are already differentiated and functional. This allows many biological questions to be addressed at this stage. For example, larvae have been useful for neuronal studies, providing a simpler model with only 10,000 neurons compared to the adult’s 250,000. The pupal stage begins after the 3rd instar larva is encapsulated and lasts around 4 days. In this stage, many larval structures are lysed. New structures are formed from imaginal disks, which give rise to adult parts like the head, legs, and wings. Some structures, like the nervous system, are preserved.
The adult fly emerges from the pupal case through a process called eclosion. The lifespan of an adult is around 30 days, but this can be variable depending on the temperature. Drosophila is considered a good genetic model partly because a female can lay a high number of eggs, around 100 per day. This generates a large progeny after genetic crosses. The flies are also noted for having very easy growth requirements in laboratory conditions. The study of adult flies has resulted in notable findings in basic biological processes. Examples of these research areas include ageing, circadian clocks, and behavioural studies.
The antennae are a pair of sensory elements located on the head part of the fly. Within the second antennal segment, there is a mass of receptor cells. This structure is called the Johnston organ. The Johnston organ is the hearing organ for the fruit fly. It permits the flies to sense different mechanosensory stimuli. These stimuli include sound, which is used during courtship, but also wind and gravity. Neuronal signalling from the antenna is sent to the brain. This signalling allows for behavioural responses to be made to these various stimuli.