Introduction
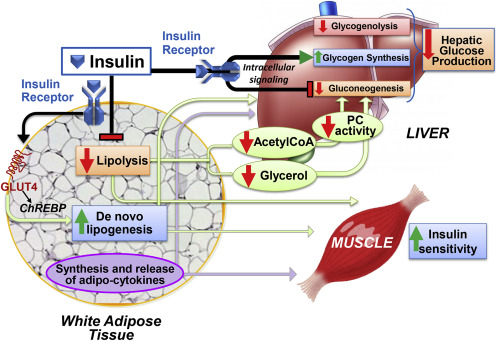
(Santoro et al) recognizes the essential importance adipocytes which react exquisitely to insulin have played in determining the mechanisms for insulin action on a cellular level on this hundredth anniversary of the discovery of insulin. The understanding of adipose remodeling has advanced significantly, and it is now clear that adipocytes are far more complicated than simple fat storage depots. A growing amount of evidence demonstrates how adipocytes contribute to the control of glucose homeostasis throughout the body in response to insulin. The review illustrates the plasticity, heterogeneity, and endocrine function of adipocytes, unique characteristics connect the metabolism of adipocytes to the regulation of other important metabolism tissues (e.g., liver, muscle, pancreas).
How to culture vascularized & immunocompetent 3D models in a standard Multiwell
Abstract of Insulin action in adipocytes, adipose remodeling, and systemic effects
In the review, the author states that “Adipocytes have been a powerhouse for understanding insulin action. While major advances have been made since the discovery of insulin 100 years ago, in defining the molecular events underlying insulin action in adipocytes and the systemic consequences, there are still many unanswered questions. We pointed out how the mechanisms for some of insulin’s effects are still unclear. This is partly because the dominant regulatory pathways for metabolic control in adipose tissue, liver, and muscle differ depending on the metabolic condition. In addition, we are just beginning to
understand how insulin action in adipocytes integrates with other inputs such as neural innervation and activation (Guilherme et al., 2019). The role of brown and beige adipose tissue in the regulation of insulin sensitivity is a very productive research area and how insulin influences the ‘‘browning’’ or ‘‘beige-ing’’ of adipocytes is of high interest. Single-cell RNA sequencing has been extremely informative for many research questions and is particularly useful for the study of adipose tissue. Going forward, a better understanding of the integrated network of cell types and pathways upstream and downstream of insulin in adipose tissue could lead to approaches to enhance systemic insulin sensitivity to prevent and improve the treatment of type 2 diabetes.”
References
Santoro, A., McGraw, T. E., & Kahn, B. B. (2021). Insulin action in adipocytes, adipose remodeling, and systemic effects. Cell Metabolism, 33(4), 748–757. https://doi.org/10.1016/j.cmet.2021.03.019
FAQ
Adipocytes are cells that show a clear reaction to insulin. They have been a powerhouse for understanding how insulin works at a cellular level. It is now clear that adipocytes are more complicated than simple depots for storing fat. Evidence shows that adipocytes are involved in the control of glucose homeostasis throughout the body, which occurs after insulin signals. The review discusses the plasticity, varied nature, and endocrine function of these cells. These specific characteristics connect the chemical processes of adipocytes to the control of other important tissues. These other tissues include the liver, muscle, and pancreas.
Major advances have occurred since insulin’s discovery in defining the molecular events of its action in adipocytes. The body-wide effects have also been studied. Despite this progress, there are still many unanswered questions. The mechanisms for some of insulin’s effects are still not clear. This is partly because the primary routes for controlling energy processing in adipose tissue, liver, and muscle can differ. These differences depend on the body’s overall condition. Additionally, our understanding is only starting to form on how insulin’s action in adipocytes combines with other inputs. An example of another input is neural innervation and activation.
The part played by brown and beige adipose tissue in the control of insulin responsiveness is a productive field of study. How insulin affects the "browning" or "beige-ing" of adipocytes is also an area of high interest. New tools are being used to investigate these questions. Single-cell RNA sequencing has been shown to provide a large amount of information for many research questions. It is considered particularly applicable to the study of adipose tissue. This method is helpful for exploring the varied nature of these cells.
A better understanding of the integrated system of cell types is needed. This includes the series of actions that occur before and after insulin in adipose tissue. Gaining this knowledge could result in new approaches. These new methods could be used to improve whole-body responsiveness to insulin. The goal of this research is to prevent type 2 diabetes. It is also intended to improve the management of this condition. The complex, varied, and endocrine functions of adipocytes connect their internal processes to the control of other tissues, which is important for these potential therapies.