Spheroids: Why using them and what it is?
In an organ, a cell needs to be considered together with its whole environment. Each cell is surrounded by a myriad of other cells, either of the same type or not. This entire environment allows cells interactions and contacts in 3 dimensions and also creates physical constraints. The classical culture cell models are for now mainly in 2D, meaning that this environment is far from the 3D environment of a cell located in an organ (other cells, extracellular matrix, interstitium…). There is a need for a 3D model with the advantage of 2D cell lines cultures (easy to do, reproducible, cheap) and approaching the 3 dimensional organization of an organ. The solution is called spheroids. This very interesting solution came out in the 1970s. Spheroids are 3D spherical organoids displaying an increase in cell density and biological functionality in comparison to classical monolayer cell culture. Spheroids are often considered as an intermediate to mimic the level of organization of a tissue/organ.
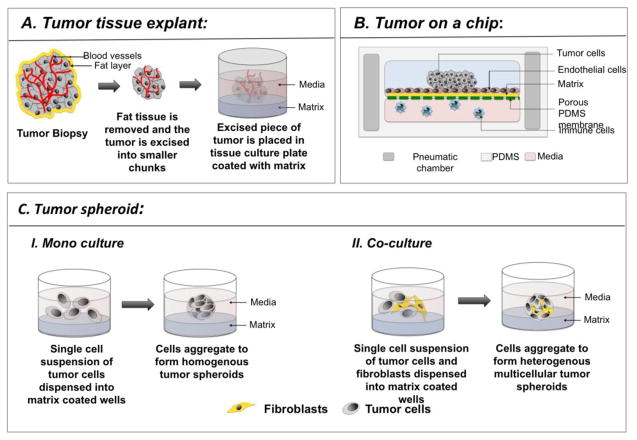
Fig 1: Application of spheroids culture in cancer research. Illustration of different ways of cultivating tumor spheroids. Adapted from Sritama et al., 2016.
Spheroids culture and tumor studies
Solid tumors are growing in a 3-dimensional architecture and possess a heterogeneous vasculature. This results in a variety of exposure to O2, nutrients, chemical agents… More generally, there is a gradient of diffusion which considerably limits the tumor response to treatments. The generation of tumor spheroids is used as an intermediate model between 2D cell culture and tumor explants culture. Spheroids may contain only one single type of tumor cell or be more complex by containing multiple cell types. Cancer researchers may be brought to measure the impact of hypoxia, oxidative stress, cancer cell metabolism, cell migration and invasion, the tumor microenvironment signaling crosstalk, and may be involved in the discovery of new anti-cancer drugs. Microfluidic chips are, for now, probably the best devices to perform such cancer research.
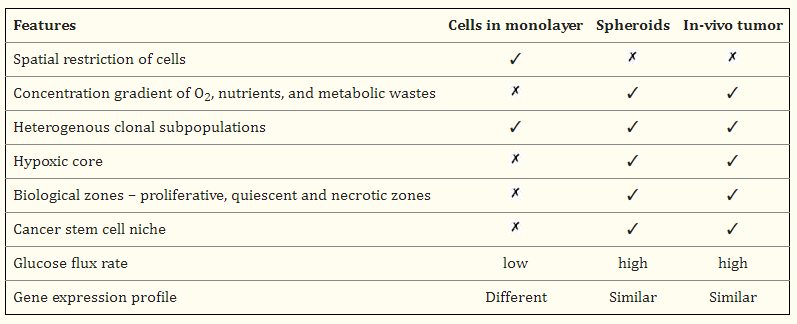
Fig 2: Key features comparison of cell monolayer, spheroids, and in vivo tumor.
How to culture vascularized & immunocompetent 3D models in a standard Multiwell
Spheroids culture and microfluidics for new Organ-on-a-chip devices
Organs-on-a-chip and more specifically, “tumors-on-a-chip” are revolutionary microengineered biomimetic models. It consists of manufacturing a functional unit of a tumor in a microfluidic device. The device allows the culture of either monocellular spheroids or the co-culture of tumor cells with other cell types in a relevant architecture that replicates the tumor microenvironment. The microfluidic device integrates a chamber allowing the attachment of the spheroids, the perfusion of the culture medium with or without a dissolved chemical.
In addition, Cherry Biotech implemented its cutting-edge microphysiological system technology to this spheroid-on-a-chip development. This is the “SpOC” project where the Cherry R&D team aims at mimicking the 3D-cellular microenvironment of an in vivo model using spheroids. The chip is composed of an inert plastic polymer, channels, and may integrate a porous membrane for advanced medium perfusion. The tumor spheroid grows in the chamber and the metastatic cells are carried away by the medium flow and might be detected afterward. Custom microfabrication of the chip and real-time data recording are some of the advantages of this technique. Real-time optical microscopy is a useful tool to follow the spheroid geometry and measure the impact of a chemical on it. Moreover, the “spheroïd-on-a-chip” model provides new avenues for genomic and drug screenings, in addition to the detection of circulating tumor cells (CTC).
References
FAQ
A spheroid is a 3D, spherical organoid. It is used as a 3D model because cells within an organ are surrounded by other cells and physical constraints. This 3D environment is not replicated by classical 2D cell cultures. A need exists for a 3D model that is reproducible and inexpensive, like 2D cultures, but also provides a three-dimensional organisation. Spheroids are considered an intermediate system. They were first developed in the 1970s. Compared to standard monolayer cell cultures, spheroids show an increase in cell density. They also display greater biological functionality. This makes them a useful step toward replicating the organisation of a tissue or organ.
Spheroids and in vivo tumors share many characteristics that are absent in 2D monolayer cultures. For example, 2D cultures grow with spatial restriction, but spheroids and tumors do not. Both spheroids and in vivo tumors show a concentration gradient of O2, nutrients, and metabolic wastes. This leads to the formation of a hypoxic core and different biological zones, such as proliferative, quiescent, and necrotic areas. These features are not found in monolayers. Spheroids and tumors also possess a cancer stem cell niche. The glucose flux rate for spheroids and tumors is high, while it is low for monolayers. The gene expression profile of spheroids is reported as being similar to in vivo tumors, whereas the profile for monolayers is different.
Tumor spheroids can be cultivated in different ways. They may contain only one single type of tumor cell or be more complex by containing multiple cell types. The diagrams show two methods for creating them. For a mono-culture, a single cell suspension of tumor cells is dispensed into matrix-coated wells. The cells then aggregate to form homogenous tumor spheroids. For a co-culture, the process is similar. A single cell suspension of tumor cells is combined with other cells, such as fibroblasts. This mixture is dispensed into the matrix-coated wells. These different cell types then aggregate together, forming heterogeneous multicellular tumor spheroids. These spheroid models are used as an intermediate system, positioned between 2D cell culture and tumor explant culture.
Spheroids are used within “tumors-on-a-chip,” which are microengineered biomimetic models. These microfluidic devices are manufactured to contain a functional unit of a tumor. The device allows for the culture of either monocellular spheroids or co-cultures of tumor cells with other cell types. This is done within an architecture that is designed to replicate the tumor microenvironment. The microfluidic device integrates a chamber for the spheroid to attach. It also allows for the perfusion of culture medium. This medium can contain dissolved chemicals for testing. The chip, which is composed of an inert plastic polymer, may also integrate a porous membrane for advanced medium perfusion.