Breast Cancer: Classification and Conventional Management
Breast cancer (BC) is defined as a complex disease characterized by high mortality and heterogeneity [2]. The incidence of the condition is influenced by sex, age, genetic background, hormone therapies, and dietary habits [2]. Conventional management of BC involves surgical intervention, chemotherapy, and radiotherapy [2]. Subtypes are categorized by the presence or absence of the estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) [2]. Triple negative breast cancer (TNBC) is defined by the absence of these three receptors and is recognized for its aggressive nature [2].
Metformin: Pharmacology and Metabolic Mechanisms
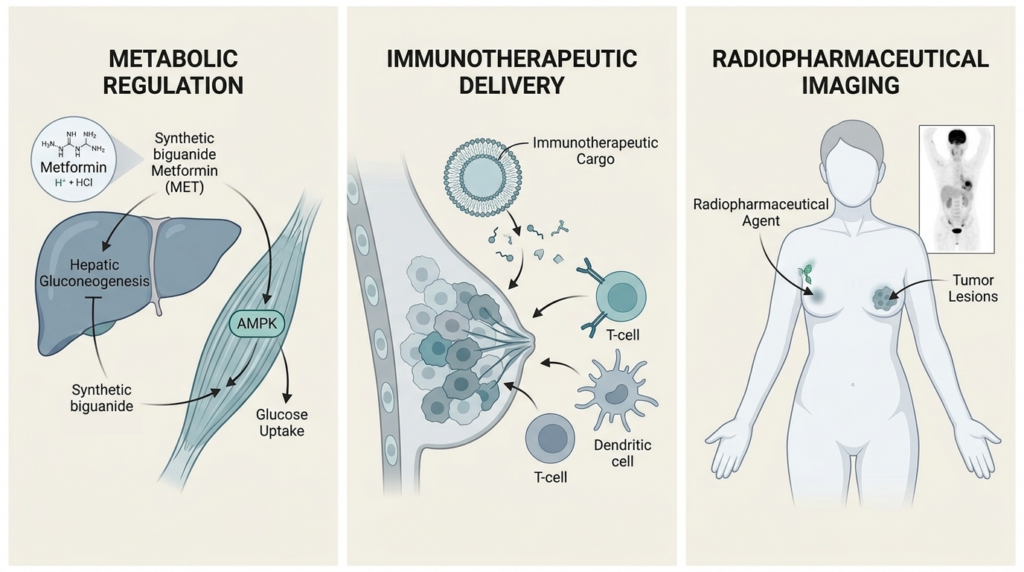
Metformin (MET) is a synthetic biguanide used for the regulation of glycaemia in type 2 diabetes mellitus (DM) [1]. Natural biguanides were previously extracted from the legume family but were associated with a risk of lactic acidosis [1]. The synthetic analogue, metformin hydrochloride, is utilized with fewer side effects [1]. MET acts as an anti-hyperglycemic agent through the activation of adenosine monophosphate-activated protein kinase (AMPK) [1]. This activation leads to the inhibition of hepatic gluconeogenesis and the induction of peripheral glucose uptake in skeletal muscles and adipose tissues [1].
Energy production in cells occurs via oxidative phosphorylation or glycolysis. Normal physiological conditions prioritize oxygen-dependent oxidative phosphorylation [1]. Cancer cells often prioritize glycolysis even in aerobic environments, a process termed the Warburg effect [1]. Glycolysis supports the synthesis of macromolecules necessary for cell proliferation [1]. Hypoxia regulatory factor 1 (HIF-1) accumulates in hypoxic conditions and induces glycolytic pathways by upregulating glucose transporter 1 (GLUT1), glucose transporter 3 (GLUT3), hexokinase (HK), pyruvate kinase M2 (PKM2), and lactate dehydrogenase (LDH) [1]. HIF-1 also inhibits mitochondrial respiration through pyruvate dehydrogenase kinase 1 (PDK1) [1].
Anti-Cancer Activity of Metformin in Breast Cancer
The anti-cancer effects of MET involve the inhibition of mitochondrial complex I in the electron transport chain [1]. This inhibition reduces cellular levels of adenosine triphosphate (ATP) and increases levels of adenosine monophosphate (AMP) [1]. The resulting increase in the AMP to ATP ratio activates AMPK [1]. Activated AMPK serves as a metabolic sensor that suppresses the growth and proliferation of cells [1]. MET is observed to attenuate the Warburg effect by decreasing glucose uptake and lactate production [1]. In BC, MET disrupts glycolytic and biosynthetic pathways driven by HIF-1 activation [1].
Cellular uptake of MET is mediated by transmembrane transporters. These include the plasma membrane monoamine transporter (PMAT) and organic cation transporters 2 and 3 (OCT2, OCT3) in enterocytes [1]. Hepatic uptake is facilitated by OCT1 and multidrug and toxin extrusion protein 1 (MATE1) [1]. Renal uptake occurs through renal epithelial OCT2 [1]. In BC cell line studies, MCF7 cells show higher levels of OCT expression and nearly threefold greater MET uptake compared to MDA-MB-231 cells [1]. PMAT expression is also reported to be threefold higher in MCF7 cells [1].
Nanocarrier-Based Delivery Systems for Metformin
Nanocarrier-based delivery systems are investigated to enhance the bioavailability and intracellular retention of MET. MET embedded in nanoparticles and radiolabelled with technetium-99m tricarbonyl core shows greater uptake in BC cell lines [1]. Nanoencapsulation using O-carboxymethyl chitosan (O-CMC) polymeric formulations is also studied for its biocompatibility in non-carcinogenic breast epithelial cells [1].
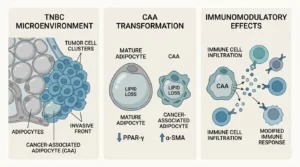
Immunotherapy and the Tumor Micro-Environment
Immunotherapy is utilized to activate the immune system of the patient to combat cancer cells, including abscopal metastatic tumors [2]. TNBC is considered an immunogenic subtype because of its high genome instability and increased mutation rates [2]. Programmed death-ligand 1 (PD-L1) is often expressed at high levels in TNBC [2]. Current strategies include adoptive cell therapy, such as tumor-infiltrating lymphocyte (TIL), chimeric antigen receptor (CAR) T-cell, and chimeric antigen receptor-natural killer cell (CAR-NK) therapies [2]. Cancer vaccines, including DNA and peptide vaccines, are also employed [2].
Immune checkpoint blockade involves targeting molecules like programmed cell death protein 1 (PD-1) or cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) [2]. CTLA-4 competes with CD28 on T cells for binding with CD80/B7-1 and CD86/B7-2 on antigen-presenting cells (APCs) [2]. Simultaneous inhibition of PD-1 and CTLA-4 by durvalumab and tremelimumab has shown clinical benefits in TNBC patients [2]. A combination of atezolizumab and nab-paclitaxel was approved by the Food and Drug Administration (FDA) for metastatic PD-L1+ TNBC [2].
Biomaterial-Based Targeting of the Tumor Micro-Environment
Nano-biomaterials are used as carrier vehicles to target the tumor micro-environment (TME) [2]. The TME contains macrophages, cancer-associated fibroblasts (CAFs), myeloid-derived suppressor cells, dendritic cells (DCs), and T lymphocytes [2]. Biomaterials are designed to release drugs in response to TME conditions like low pH and hypoxia [2]. The combination of trastuzumab and HER2 vaccines has been studied for HER2+ BC patients [2].
Radiopharmaceutical Imaging with FDG-PET
Positron emission tomography (PET) is a functional imaging technique used to evaluate cancer pathways [3]. 2-deoxy-2-[18F]-fluoro-d-glucose (FDG) is the most common radiopharmaceutical used to investigate glucose metabolism [3]. FDG-PET is recommended by European Society of Medical Oncology (ESMO) guidelines for the staging of suspected oligometastatic disease [3]. It shows diagnostic accuracy comparable to magnetic resonance imaging (MRI) for detecting bone metastases [3].
Limitations of FDG-PET include a spatial resolution limit for lesions smaller than 5 mm [3]. Physiological FDG uptake in the brain prevents the evaluation of central nervous system metastases [3]. BC heterogeneity also affects FDG avidity. Invasive lobular cancers show lower uptake than invasive ductal cancers [3]. Tumors with low Ki-67 levels also show reduced FDG avidity [3].
Novel PET Tracers and Theranostic Approaches
Other PET radiopharmaceuticals are designed to target specific tumor characteristics. FluoroEstradiol (FES) is an FDA-approved tracer for targeting the estrogen receptor (ER) in metastatic BC [3]. FES-PET helps evaluate ER heterogeneity and the efficacy of hormonal therapy [3]. Inhibitors of the fibroblast activation protein (FAPi) are used to image CAFs in the TME [3]. FAPi is also proposed as a theranostic agent for radio-ligand therapy [3].
Tracer agents like [89Zr]Trastuzumab and [64Cu]DOTA-Trastuzumab target HER2 [3]. These tracers assist in clinical decision-making when HER2 expression cannot be assessed by biopsy [3]. [89Zr]Trastuzumab shows high tumor to background ratio (TBR) in the liver, spleen, kidneys, and brain [3]. Integrin αvβ3 expression is evaluated using [18F]Galacto-RGD [3].
References
- Jinadasa AGRG, Wageesha NDA, Samarakoon SR, Ekanayake S, Akalanka HMK. Metformin-Mediated Glycaemic Regulation as a Potential Strategy for Breast Cancer Prevention. PubMed. 2026.
- Santerre JP, Yang Y, Du Z, Wang W, Zhang X. Biomaterials’ enhancement of immunotherapy for breast cancer by targeting functional cells in the tumor micro-environment. PubMed. 2024.
- Gilardi L, Airò Farulla LS, Ceci F. Imaging for illuminating actionable pathways in breast cancer. PubMed. 2022.
FAQ
HER2-positive breast cancer accounts for approximately one-quarter of breast malignancies. This cancer type is driven by the amplification of the ERBB2 gene. Although targeted agents are used, resistance to treatment and varied therapeutic responses remain serious problems. Traditional research methods, such as two-dimensional cultures and xenografts, have provided molecular information. These older models cannot, however, reproduce the intricate structural and signaling organization found in human tumours. New experimental systems were developed to better represent human tumour physiology. These systems include organoids and breast cancer-on-chip devices. They are intended to provide more applicable avenues of research into HER2 signaling and drug responses.
HER2-positive organoids provide experimental systems that are considered more physiologically relevant than conventional cultures. These models, which include cell-line derived and patient-derived organoids, maintain features of the original tissue. These features include native polarity, interactions with the extracellular matrix, and intratumoral heterogeneity. This structure permits analysis of HER2 signaling dynamics. It also allows study of drug penetration within realistic gradients. Microfluidic breast cancer-on-chip approaches are used to integrate vascular and immune components under controlled flow. These systems support studies on antibody delivery, immune cell engagement, and adaptive resistance. By combining genetic fidelity with microenvironmental control, these methodologies are altering preclinical HER2 research.
Cell-line derived organoids are used to study how tumour architecture affects signaling. Recent analyses compared BT474 and SKBR3 organoids, as both are HER2-positive lines. Large differences in tissue organization and receptor signaling were found. BT474 cells were observed to form compact, spherical aggregates. These structures had defined polarity and reduced AKT phosphorylation. In contrast, SKBR3 cultures developed loosely organized structures. These showed increased HER2 expression but diminished cohesion. Confocal microscopy revealed that HER2 receptor density decreases toward the center of larger aggregates in both models. These morphological differences show how structural organization modulates HER2 activity. Tumour geometry and cell-to-cell contact appear to affect oncogenic signaling.
Drug screening studies show that antibody-based therapies behave differently in a three-dimensional architecture. The cytotoxic effect of a HER2-directed immunotoxin was tested. Its effect was found to be several orders of magnitude lower in HER2-positive cell-line derived organoids compared to 2D cultures. This difference was attributed to limited diffusion. The compound was restricted to the outer layers of the organoid, so central cells were largely unaffected. Similar findings were reported for trastuzumab. Microfluidic devices that generate linear drug gradients showed large differences in concentration-dependent responses. These observations suggest that reduced drug penetration may be a reason for resistance seen in clinical settings. This is particularly true in dense tumour regions that have poor vascularization.
A “tetraculture” model is a type of multicellular organoid. It is designed to incorporate stromal and immune components, which expands the predictive power of cell-line derived organoids. This model combines HER2-positive epithelial cells with fibroblasts, macrophages, and endothelial cells. The resulting structures were found to recapitulate patient-specific tissue organization. The localization of fibroblasts and macrophages differed depending on the tumour subtype. As an illustration, in HER2-positive BT474 organoids, cancer-associated fibroblasts aggregated in the center. This created compact structures that contained hypoxic cores. In contrast, SKBR3 organoids showed a more dispersed stromal pattern and peripheral macrophage accumulation. These different arrangements produced spatial gradients in viability and metabolic activity.
Patient-derived organoids (PDOs) are established directly from patient tissue, including metastatic biopsies. A main feature of these models is that they preserve the histological and molecular identity of the original tumour. This includes ERBB2 amplification, hormone receptor status, and other genomic alterations. PDOs provide an opportunity to observe patient-specific tumour behaviour under controlled experimental conditions. They also maintain the genetic diversity that defines clinical heterogeneity. Organoid biobanks have shown that these cultures retain the architecture, receptor expression, and growth characteristics of their tissue of origin. In addition, PDOs have been shown to maintain genomic stability across passages, which makes them suited for longitudinal analysis.
Patient-derived organoids are used for direct assessment of therapeutic sensitivity on tissues from a patient. This provides a route to individualized treatment design. One 2022 study used organoids to guide therapy in a metastatic case, achieving a complete clinical response. These models are also used to study resistance. Organoids grown from tumours that had lost HER2 after therapy were analyzed. These cultures required neuregulin-1 for growth, which revealed adaptive signaling changes. This provided a model to evaluate HER2-independent treatment strategies. PDOs also permit combination therapy evaluation. Studies using organoids from HER2-low and HER2-amplified tumours compared different drugs. Although trastuzumab alone often had limited cytotoxicity, adding pan-HER inhibitors like neratinib increased growth inhibition and delayed recovery.
A main advantage of microfluidic breast cancer-on-chip systems is the ability to reproduce vascular perfusion. This creates a situation that reflects drug transport and clearance. One study developed a chip where breast cancer–fibroblast organoids were enclosed by self-assembled human endothelial networks. Chemotherapeutic agents were delivered through these perfused microvessels. Under flow conditions, cytotoxic responses were observed to be markedly weaker and more gradual than in static cultures. This finding suggests reduced overall drug accumulation within the tumour tissue. Another study reported diminished cancer cell death under continuous perfusion, even with equivalent drug concentrations. These studies show how fluid dynamics affect therapeutic exposure. Conventional static assays may overestimate efficacy.
Microfluidic chips provide fine spatial control, which is used to study interactions between tumour cells and stromal components. One design used a chip with two adjacent chambers separated by microscale pillars. One side was seeded with MCF-7 cancer cells and the other with normal fibroblasts. This porous divider permitted cell-to-cell contact and molecular crosstalk. It was shown that co-culture with tumour cells induced fibroblast activation to a pro-tumorigenic myofibroblast state. This process resembles the desmoplastic reaction seen in actual tumours. Other chip systems are used to examine cancer cell migration and invasion in real time. A central region holds a spheroid of tumour cells embedded in matrix, while adjacent channels contain collagen or stromal cells. Investigators can monitor tumour cells invading out of the spheroid and quantify how different conditions affect this invasion.
Yes, both patient-derived organoids and chip systems are used for immunotherapy research. HER2-expressing PDOs have been co-cultured with chimeric antigen receptor (CAR) natural killer (NK) or T cells. In these experiments, the engineered immune cells successfully penetrated the organoid matrix. They also selectively eliminated HER2-positive cells. This method shows measurable antitumor efficacy and cytokine release patterns. Breast cancer-on-chip platforms are also used. Systems combining vascularized HER2-positive tumour organoids with a perfusable endothelial interface have been developed. This setup allows for real-time tracking of immune cell extravasation as cells traverse the endothelial layer. It is used to evaluate the effectiveness and selectivity of HER2-directed CAR-T and CAR-NK cells in a human-specific context.